Therapeutic Peptides are a class of pharmaceutical agent composed of a series of well-ordered amino acids usually having the molecular weight between 500-5000 Da.1 The discovery of several lifesaving bioactive peptides like the 51-amino acid hormone insulin,2 and the 39-amino acid adrenocorticotrophic hormone are considered important scientific achievements in drug discovery. A good peptide editor is one of the important tools for researchers and scientists working in the field of peptide design and drug development.
The peptide editor in Flare allows researchers to design and modify peptide sequences. It provides a user-friendly interface to input amino acid sequences and visualize the resulting peptide. This is crucial for designing peptides with specific functionality such as binding to a target protein or exhibiting therapeutic properties. Additionally, the peptide editor helps users to optimize several properties of peptides like solubility, stability, and bioavailability. By adjusting the sequence or modifying specific amino acids, researchers can enhance desired characteristics and minimize undesirable ones.
How to use the Peptide Editor in Flare
The peptide editor option is available in the ‘Editing’ tab in Flare. The ‘Grow Peptide’ button contains the peptide editing functionality. Selecting the ‘Grow Peptide’ button will open a pop-up window where the amino acid sequence of the desired peptide can be typed or pasted. The amino acid is depicted using the 1-letter code. If a new editing session is started the peptide is created as an independent fragment; if a protein structure or an amino acid sequence is instead being edited, and the current selection includes the C-terminus, then the peptide is appended to the terminus. In the example below, the single-letter peptide sequence of Met-enkephalin, a naturally occurring, endogenous opioid peptide is typed as ‘YGGFM’ where Y: Tyrosine, G: Glycine, F: Phenylalanine, and M: Methionine. The peptide is created as an extended conformation.
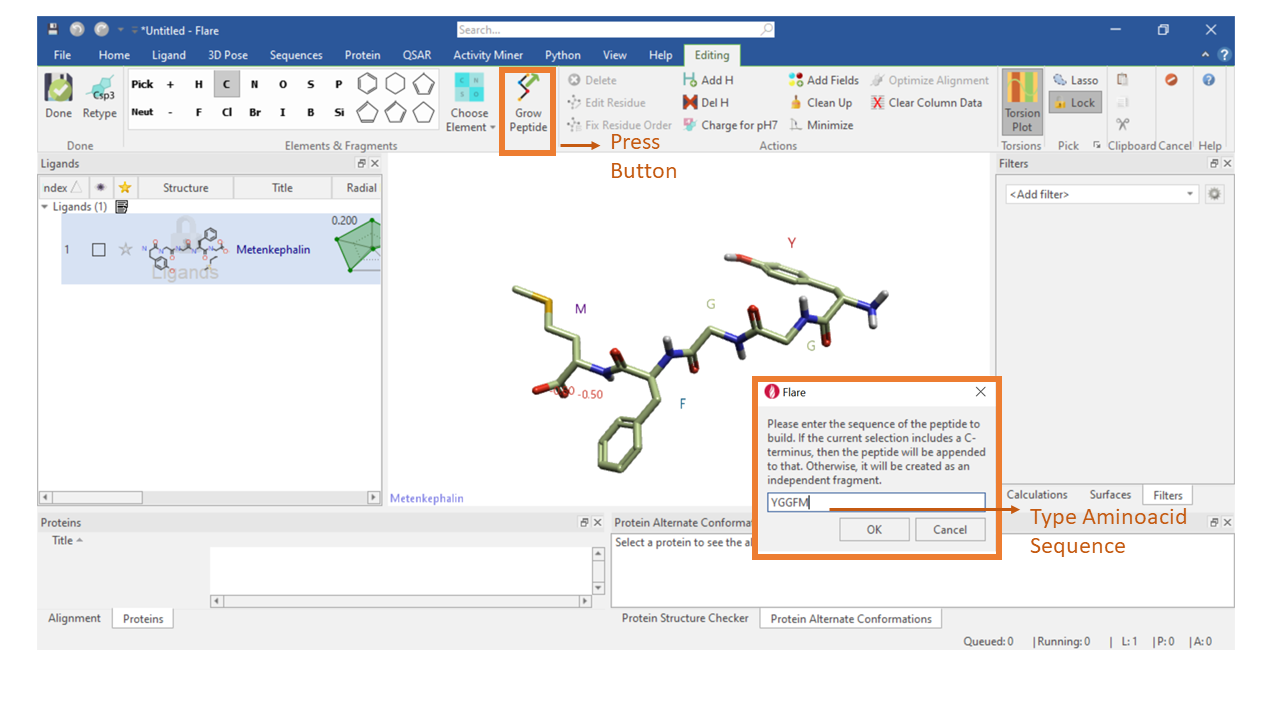 Figure 1. Example of editing a peptide using the ‘Grow Peptide’ button in the editing tab of Flare.
Figure 1. Example of editing a peptide using the ‘Grow Peptide’ button in the editing tab of Flare.
An example of building peptide-based ligands
A ligand can also be generated by the combination of both the Peptide editor and element fragment editor. As an example, we have chosen to draw aspartame, a methyl ester of the aspartic acid phenylalanine dipeptide that is used as an artificial non-saccharide sweetener.
Select ‘Grow Peptide’ in the editing tab, then type the dipeptide sequence “DF”, where D: Aspartic acid and F: Phenyl alanine. Now use the element editor, to edit carboxylic acid group of the C-terminus to a methyl ester group. Click the ‘Clean up’ button to optimize the bond parameters and rename the new ligand as aspartame.
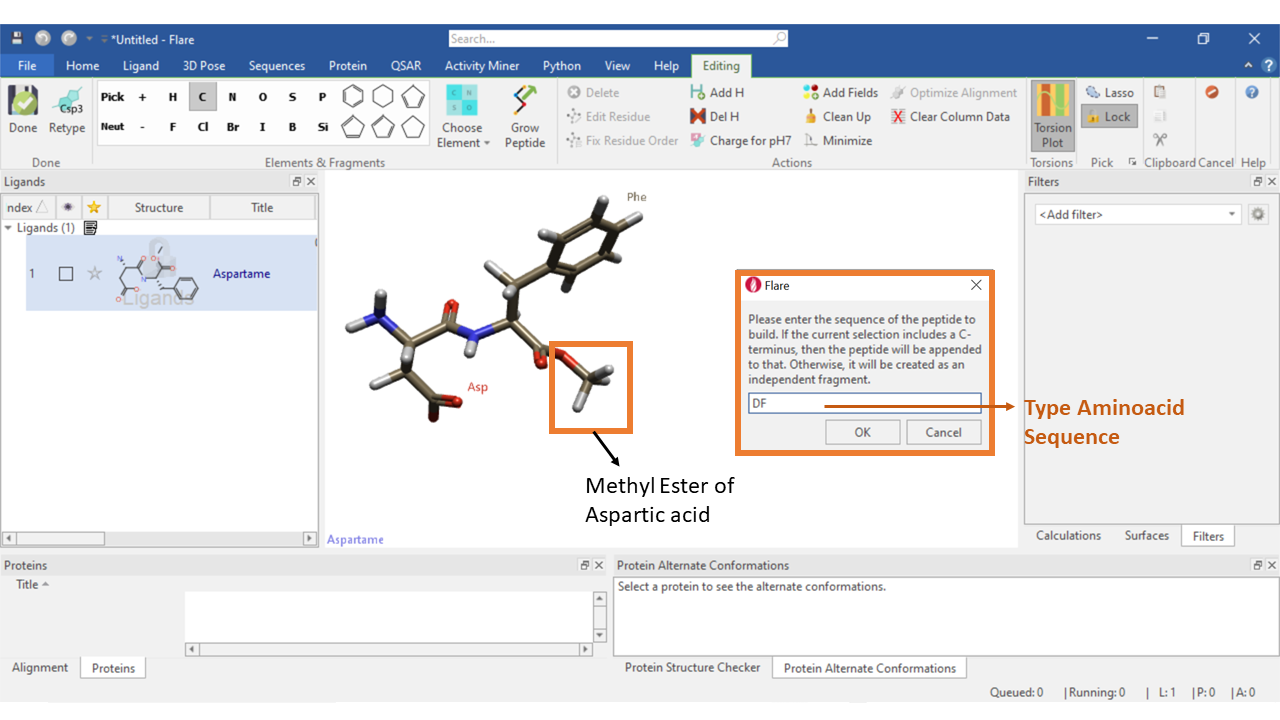 Figure 2: Building an aspartame molecule using a combination of peptide growing tool and element editing tool in Flare.
Figure 2: Building an aspartame molecule using a combination of peptide growing tool and element editing tool in Flare.
If you’d like to take the opportunity to explore Flare and its full portfolio of CADD features, then please contact us today to request a free evaluation. As part of the evaluation process, you’ll receive full support in installing the platform and accessing its wide range of features, while having the freedom to publish any results produced and use these for further research.
References
1. Henninot, A., Collins, J. C. & Nuss, J. M., The current state of peptide drug discovery: back to the future? J. Med Chem. 61, 1382–1414 (2018).
2. (a) Bliss, M., Banting’s, Best’s, and Collip’s accounts of the discovery of insulin, Bull. Hist. Med 56, 554–568 (1982). (b) Scott, D. A. & Best, C. H., The preparation of insulin, Ind. Eng. Chem. 17, 238–240 (1925).