Design-Make-Test-Analyze
Irrespective of the therapeutic area or target pathway, all small molecule discovery teams repeat a common workflow to optimize their identified hits towards clinical candidates: the Design-Make-Test-Analyze (DMTA) cycle.
Figure 1: The Design-Make-Test-Analyze cycle drives productivity in drug discovery.
The time it takes to complete each DMTA cycle is a major factor in overall project productivity. While efforts have gone in to optimizing the drug discovery process, there are further opportunities for improvement. Fractured workflows can result in a distinct lack of coordination across a project and this is even further compounded when companies turn to outsourcing.
Effective communication is critical under fragmented working conditions
Each step of the DMTA cycle is dependent upon the output of the other three and so cycle time relies heavily on effective communication between all parties involved. Yet still, a truly collaborative approach is difficult to coordinate and progress updates are often reserved for timetabled meetings or lost in a deluge of emails. In reality, decisions are made every day and, unless there is coordination with the rest of the team, these can result in wasted time and duplicated effort. A chemist may invest time working on a design that has already been attempted and discarded, or two chemists may even be unknowingly working on the same design simultaneously.
Lack of communication and insufficient visibility on a project can significantly hinder productivity and ultimately cost time and money which hinders progress towards the clinic. COVID-19 induced disruptions have heavily impacted the way research teams work, meaning it is now commonplace for scientists to work fragmented shift patterns, or from remote locations. As a result, it is more challenging than ever for managers to have a full overview of the project and for team members to be kept up to date with the latest developments. Coordination is vital to ensure good resource management and prevent unnecessary delays in project progression.
Torx™ promotes collaborative working and information delivery
Scientific discovery is a dynamic process, which Torx™ accommodates. This innovative platform acts as a comprehensive information delivery agent, accessible to all team members to review and input on the latest updates in real time, throughout all stages of the DMTA cycle. For example, synthetic chemists can have visibility on designs as they are being developed, which enables them to inform on prioritization based on their experience and insight. Valuable time can be saved by removing the need for laborious reporting so that meetings can focus solely on strategic discussions around project direction.
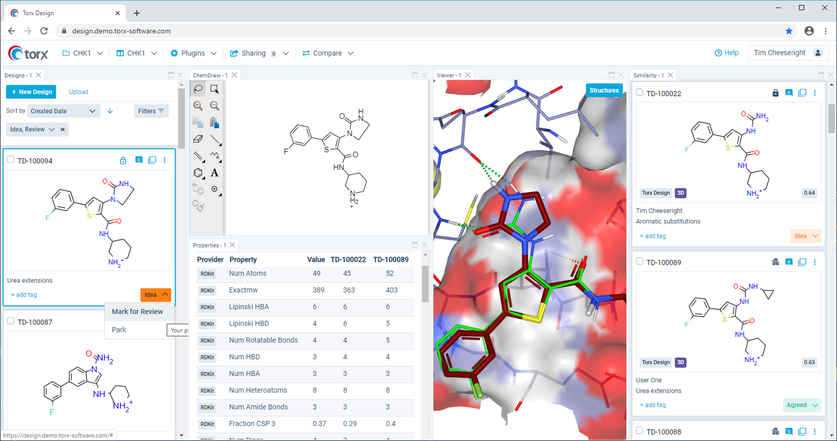
Figure 2: Torx gives enhanced visibility throughout a project, enabling all team members to input on design prioritization.
Requirements may change with growth
Although challenges surrounding effective communication and resource management are ubiquitous, drug discovery companies may have specific requirements depending on their size and the level of internal resources and expertise.
Case A: Small / start-up biotechnology companies
Consider a biotech company in the very early stages of its development. In order to test and confirm its hypothesis, the leadership team may opt to outsource all DMTA areas rather than risking large investments in developing internal expertise. Communication becomes increasingly difficult as more parties are involved so it is vital for a successful output of the project to bring everyone together on the same page as quickly as possible.
Torx offers an efficient information flow and has the ability to connect disparate teams and services. The level of visible information can be custom defined so that the workflow can be shared with external collaborators without compromising confidentiality. Torx Software can offer a completely outsourced IT solution, by hosting both the Torx platform and the back-end systems for a seamless end-to-end experience.
Case B: Large / multinational pharmaceutical companies
At the other end of the spectrum, large multinational pharmaceutical companies have very different needs. Over its lifetime, a company will have amassed a vast amount of data and will likely have created numerous repositories for different areas of the DMTA cycle. However, this approach can be cumbersome and an inefficient way of working. It can be very difficult to find desired information quickly when there are multiple libraries to search through. This information may be vital to support patent applications, freedom to operate assessments or project progression deadlines and the lack of a holistic solution could cause significant delays to productivity.
In this case, Torx, as a web-based platform, can be fully integrated into legacy systems to provide a single, streamlined solution for information delivery. Data integrity, internal and external connections will be ensured, whilst providing a smooth user experience.
Case C: Everything in between
Of course, in reality, a large percentage of companies will fall somewhere in between the two scenarios. They may have chosen to outsource some areas of the business and developed some in-house solutions to manage their increasingly complex data. It is here that the flexibility of Torx can be exploited to best align with specific corporate requirements, while maintaining an easy end-user experience.
Final considerations
Improving DMTA cycle efficiency is a main focal point of small molecule discovery programs today and could reduce the time and costs involved in lead discovery and optimization. Torx is a re-imagined scientific workflow, that captures all information throughout the DMTA cycle. Independent of the therapeutic area or the company size, Torx can be integrated into your drug discovery workflow, to inspire collaborative working and drive productivity.
Streamline your DMTA cycle
Contact us to request an evaluation of Torx or for a discussion about inspiring your discovery chemistry teams to work together faster.