
Background on Serialization
The Drug Supply Chain Security Act (DSCSA) requires pharmaceutical trading partners to share data that tracks products and provides traceability through the supply chain. The sharing of serialization data relies on Electronic Product Code Information Services (EPCIS), an internationally accepted GS1 standard. EPCIS supports interoperable exchange of serialization data between trading partners.
Today, DSCSA only requires sending lot-level transaction data. However, full traceability requirements mandate EPCIS data exchange at the serialized package level by November 27, 2023. This will require cooperation between manufacturers, wholesale distributors, service providers and other supply chain members. The increased traceability is ultimately intended to enhance U.S. pharmaceutical supply chain security.
Accelerating the Serialization Timeline
AmerisourceBergen, Cardinal Health and McKesson recently addressed these requirements proactively.
- Read the paper from AmerisourceBergen here.
- Supplier letters stating EPCIS data will be required by November 2022. This requirement includes their traditional wholesale distribution as well as specialty distribution arms and is driven by the need to work through issues in collecting serialization data and sending it on to dispensers. Read the letter from Cardinal Health here.
Other distributors will likely follow their lead. Specialty pharmacy providers and other dispensers may also begin demanding data in advance of the 2023 enforcement deadline.
What Biopharmaceutical Manufacturers Need to Know
For companies using Third-Party Warehousing (3PL) partners, there are several options for establishing compliant capabilities. However, manufacturers will continue bearing the regulatory responsibility to affix serialization data to packages and homogenous cases, send serialization data to customers, store/maintain serialization records, and respond to product verification requests. This responsibility will stay with the manufacturer regardless of who performs packaging, facilitates serialization data exchange, ships physical product, or performs other related activities on behalf of the manufacturer.
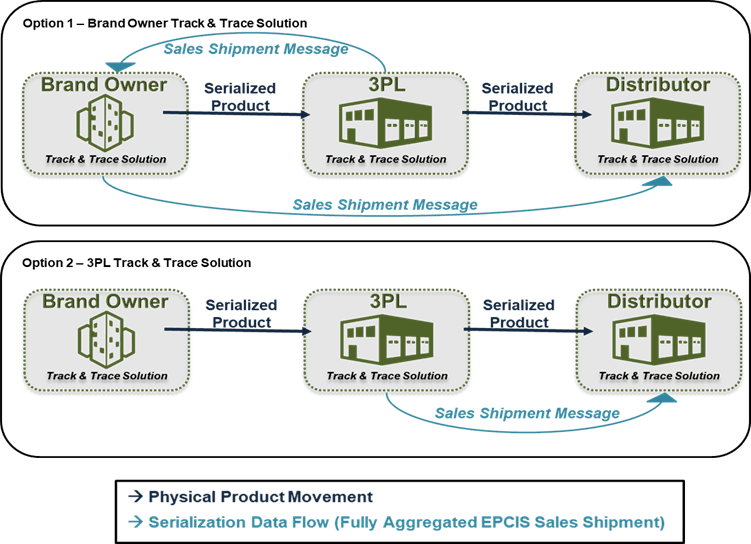
Manufacturers May Send EPCIS Data Themselves
Manufacturers may use their own serialization solution and resources to send data downstream to their distributors. This includes capturing data by scanning serialized barcodes and compiling the aggregated data for shipment. This can be accomplished regardless of whether warehouse operations and scanning are performed in-house or at a 3PL. Manufacturers should work with their serialization solution provider to identify and set up the functionality needed to capture and send EPCIS data to distributors if they have not already.
Manufacturers May Contract with Their 3PL Partner for EPCIS Data
When manufacturers contract with 3PLs for warehousing, logistics, and/or order fulfillment services, the 3PL also performs physical shipment of product to customers. Many 3PLs are establishing capabilities to capture serialization data for shipments during outbound scanning, and depending upon those capabilities, manufacturers may be able to leverage their services to send EPCIS data to distributors.
For a detailed outline of the costs and risks manufacturers should consider while evaluating their 3PL options, check out 2022 Industry Serialization Requirements: Assessing 3PL Options for Sending EPCIS Data to Distributors
The full traceability requirements stand regardless of whether the manufacturer is already serializing commercial products or is planning a commercial launch in 2022.
Immediate Next Steps
To meet Cardinal Health’s and McKesson’s serialization requirements by November 2022, manufacturers should take the following steps now:
- Ensure you have the capability to perform serialization data aggregation at the time of packaging – In previous stages of DSCSA enforcement, aggregation has always been a future requirement, but the time for aggregation is now. Unless already in place, contact your packager to plan for establishing aggregation capabilities. This may require installing and qualifying new equipment, which can take months to complete.
- Understand your 3PL’s offerings and capabilities – Meet with your 3PL to learn their process for outbound scanning and sharing EPCIS data for customer shipments. Be sure to clarify the available options for sending the data directly to distributors and/or to your own serialization management/track & trace solution.
- Evaluate the functionality of your serialization solution software – Contact your solution provider to understand what additional functionality you may need, whether you currently own/are subscribed to it, and what it will require to implement. The software provider can give you a cost estimate to access and implement any missing solution functionality.
- Perform a risk and cost analysis – Conduct an assessment based on your own unique business risks and costs, then determine a path forward. Be sure to include those costs in your 2022 budget!
On an industry level, these efforts will strengthen the security of the drug distribution supply chain. However, there is limited expertise when it comes to implementing solutions to meet DSCSA requirements. Collaborating with external partners and service providers who have experience evaluating and implementing these solutions can extend manufacturers’ internal bandwidth, promote efficiency and effectiveness in these efforts. It can also provide assurance that the implemented solutions are the right fit for the organization.
About

Stephanie L Byrne
Serialization Lead at Converge Consulting
Stephanie L Byrne is a Senior Supply Chain Consultant and Serialization Lead focusing on DSCSA project leadership. She has helped more than a dozen life sciences companies realize the greatest impact from their serialization projects by bridging the gap between sound strategy and reliable execution.